Our quality systems are designed as per ICHQ Guidelines
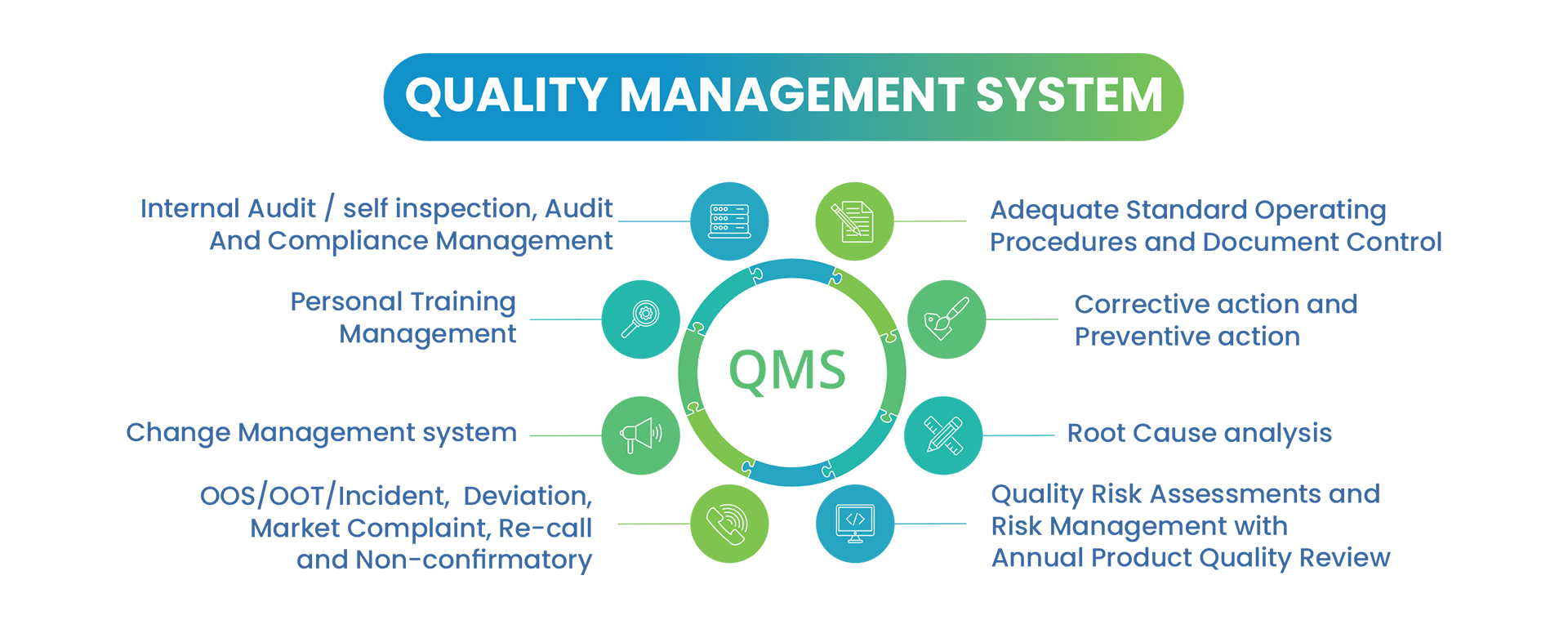
Quality Assurance is responsible for the site-specific maintenance, compliance, realization, and further development of internal Quality Management Systems. Quality Assurance ensures that APIs and advanced intermediates produced from various stages of manufacturing to comply with cGMP requirements. Following are a few of the important activities handled by the Quality Assurance team.
Our vision is to achieve international standards of quality, through persistent hard work and better implementation of knowledge by the people and have a corporate quality policy in place. Our quality systems are designed as per ICHQ Guidelines and to implement these standards we have a thorough training and re-training program in place.

